Herpes virus membrane fusion requires multiple viral proteins to participate in multiple cell surface receptors, and the coordination process can be completed. The whole process is extremely complicated. Identification of viral surface glycoprotein D (gD) and host cell receptors is an essential step in the early stages of alpha-herpesvirus infection. Among the gD receptors identified so far, the cell adhesion molecule nectin-1 is involved in the process of invasion of host cells by various α-herpesviruses and is considered to be the most effective gD receptor. Therefore, gD recognizes the molecular mechanism of nectin-1 as an important scientific issue in the field of alpha-herpesvirus research.
The Gaofu team and the Yan Jinghua team of the Institute of Microbiology, Chinese Academy of Sciences have analyzed the complex structure of herpes simplex virus (HSV) gD and its receptor nectin-1 in the previous study, and found that HSV gD protein binds to nectin-1. The contact surface of the molecule dimerization. This mode of binding disrupts the dimerization of nectin-1 itself, thereby impairing cell adhesion and facilitating viral invasion (Nature Communications 2011; Journal of Virology 2014). It also elucidates the interaction mechanism of another key glycoprotein, gB, of the HSV virus with the receptor PILRa (PNAS 2014).
Pseudorabies virus (PRV) is the same as herpesvirus in HSV. HSV is a member of the Simplexvirus genus and PRV is a member of the Varicellovirus genus. Studies have shown that PRV can also use nectin-1 as a receptor, however its molecular mechanism is unclear. In addition, if the PRV infected host is a pig, can it bind to HSV receptors, such as human nectin-1, HVEM, etc., and there is a risk of infecting humans?
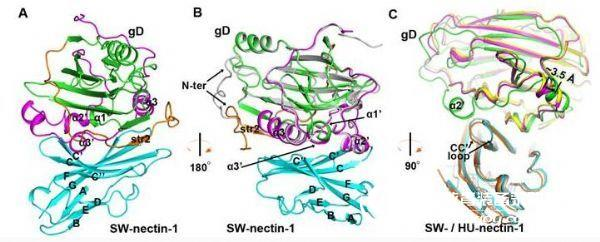
The team systematically studied the interaction of PRV gD with nectin-1, demonstrating that PRV can infect human and porcine-derived nectin-1 overexpressing cells, and PRV gD has essentially the same affinity for nectin-1 from both sources, a warning People must guard against the risk of PRV infection. The study further analyzed the extracellular domain of PRV gD and its crystal structure with nectin-1 complex. The structure shows that the spatial structure of PRV gD is very similar to HSV, although its sequence homology is only 22%. Unlike the HSV gD structure, the loop region of the N-terminus of PRV gD is relatively short, and this region is just HSV gD combined with another The location of a receptor HVEM, which explains why PRV cannot use HVEM as a receptor. The complex structure indicates that PRV gD binds to nectin-1 in the same pattern as HSV. However, there are several unique features at the interface between PRV gD and nectin-1, which promote PRV ligands to utilize amino acids different from HSV gD. The residue interacts with nectin-1. This study not only reveals the molecular mechanism of interaction between PRV gD and nectin-1, but also enriches the understanding of the receptor binding pattern of the alpha-herpesvirus subfamily, and also develops small molecule drugs that inhibit the fusion of pseudorabies virus. Lay the theoretical foundation.
The research was recently published in the journal PLOS Pathogens. Li An, a doctoral student of the Institute of Microbiology of the Chinese Academy of Sciences and Guangxi University, and Yan Guangwen, a professor at Sichuan University, shared the first author, Gao Fu, Yan Jinghua, and Professor Luo Tingrong of Guangxi University as co-authors of the paper. The research was funded by a national key R&D program and several National Natural Science Foundation projects.
Office Chair,Ergonomic Executive Office Chairs,Adjustable Chair Office,Swivel Office Chair
Ruian Jia Beir Auto Parts Co.,Ltd. , https://www.jbrseat.com